The Role — and the Risks — of Compounding Pharmacies
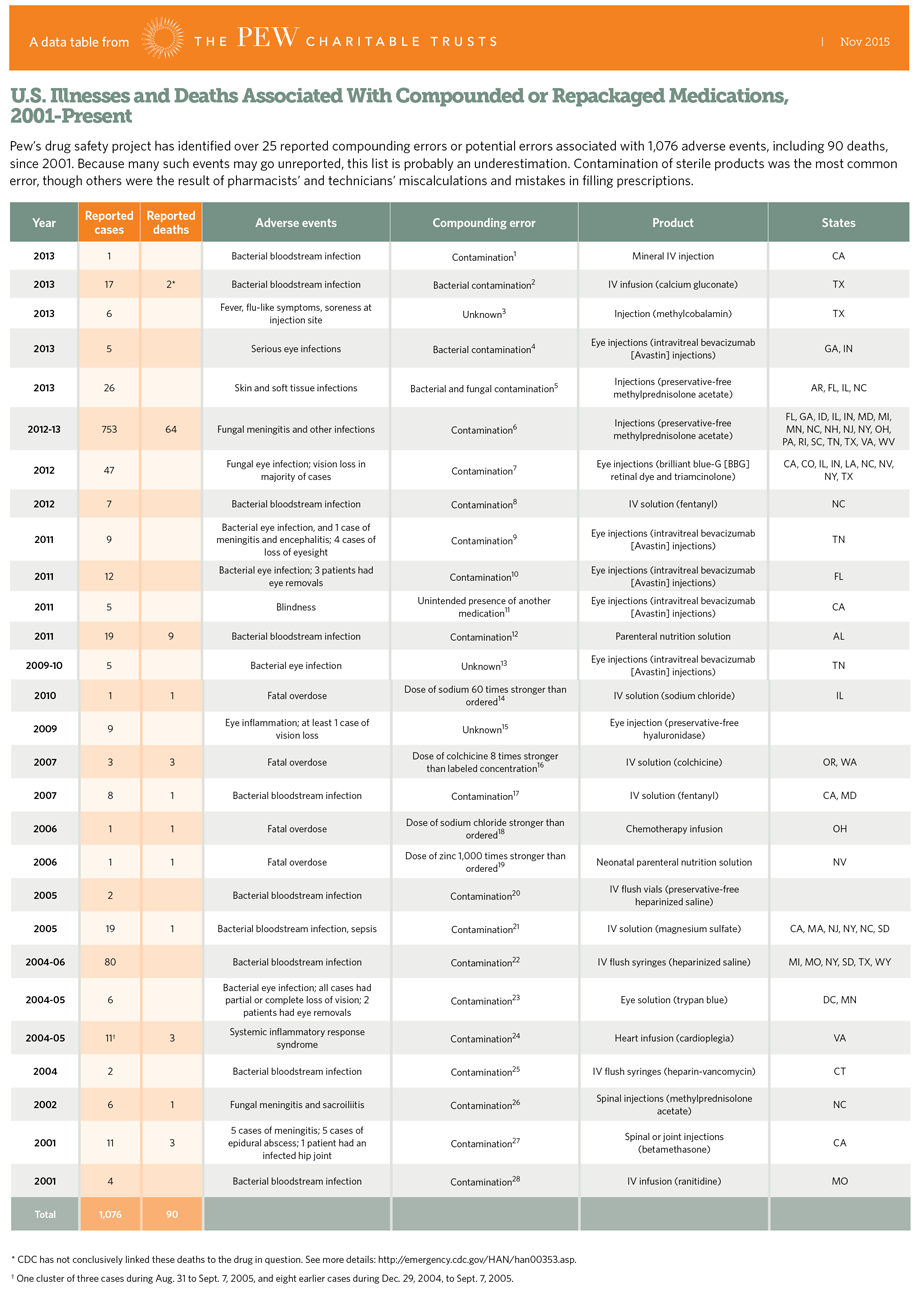
Compounding pharmacies play a valuable role when patients need a custom medicine — such as a unique dose, liquid form, or ingredient change — or when drug shortages leave no other option. But, unlike manufacturers, compounding pharmacies don’t have to meet the same strict FDA standards for testing, safety, or quality control. As a result, the quality and safety of compounded drugs may not be consistent or fully reliable.
For these reasons, their use should be limited to patient-specific preparations and adding emergency supply during drug shortages. Using them as a shortcut for lower prices or supply can put patients at risk and undermine confidence in medicines produced under the proven FDA standards that ensure medicines are both safe and effective.

ASBM Letters in Support of SAFE Drugs Act of 2025
ASBM Statement on FDA Commissioner’s Comments Regarding Compounded Medicines
The Alliance for Safe Biologic Medicines (ASBM) welcomes FDA Commissioner Marty Makary’s comments on the social media platform X affirming that “the FDA cannot verify the quality, safety, or effectiveness of non-approved drugs” and warning that the agency will “take swift action against companies mass-marketing illegal copycat drugs.” These remarks accurately reflect ASBM’s longstanding concern regarding large-scale compounding pharmacies that copy FDA-approved medicines while sidestepping the FDA’s rigorous safety, quality, and oversight standards that protect patients.
Legislation

Senator Jim Banks Introduces SAFE Drugs Act to Crack Down on Risky and Fraudulent Weight Loss Drugs
This week, Senator Jim Banks (R-Ind.) introduced the Safeguarding Americans from Fraudulent and Experimental (SAFE) Drugs Act. The bill would strengthen existing guardrails and close loopholes in the Food, Drug, and Cosmetic Act to stop unapproved compounders from mass-producing risky drugs in unregulated and uninspected facilities. Senator Martin Heinrich (D-N.M.) cosponsored the bill.

SAFE Drugs Act of 2026
Congressman Rudy Yakym (R-IN-02) and Congressman André Carson (D-IN-07) introduced the “Safeguarding Americans from Fraudulent and Experimental (SAFE) Drugs Act of 2025” to protect patients from untested, unapproved, and potentially dangerous mass compounded drugs.
News & Articles
National Consumers League Commends FDA for Swift Action Against Hims & Hers
National Consumers League (NCL)
Media Contact: Lisa McDonald, Vice President of Communications, 202-207-2829

Tom Cotton demands FDA probe into illegal Chinese ingredients in US weight loss drugs
By Alex Miller | Fox News

UPDATE: FDA's Makary pledges crackdown on mass marketing of 'illegal copycat drugs' in wake of Hims' Wegovy pill push
Fierce Pharma | By Fraiser Kansteiner

GLP-1 Compounders Endangering Americans
DC Journal | John Hertig

PSM statement on ‘deeply concerning’ compounding of GLP-1 weight-loss pills
Partnership for Safe Medicines

Injecting Clarity: The State of Medical Spa Regulation in the US
John B. Hertig, PharmD, MS, CPPS, FASHP, FFIP, Hertig Healthcare Advising, LLC

Moving the Needle: A Joint Enforcement Operation Against Improperly Licensed Medspas in NYC
The New York City Council Oversight and Investigations Division
Prescription Drug Freight Fraud Report, November 2025
The Partnership for Safe Medicines

Compounding Pharmacies Are Not a Solution to High Drug Costs
DC Journal - Philip J. Schneider and Ronald P. Jordan | March 24, 2025
Compounding Without Safeguards: A Prescription for Risk
Washington Times | By Philip J. Schneider & Ronald P. Jordan
Resources

ASOP Foundation 2025 Consumer Behavior Survey
The ASOP Global Foundation conducted this survey to assess how Americans perceive, purchase, and evaluate the safety of prescription medicines sold online.

INTELLIGENCE ALERT:
Distribution of unregulated, uninspected, unsanitary and potentially unsafe diabetes and weight loss injectables through unlicensed med spas poses immediate threat to public safety.
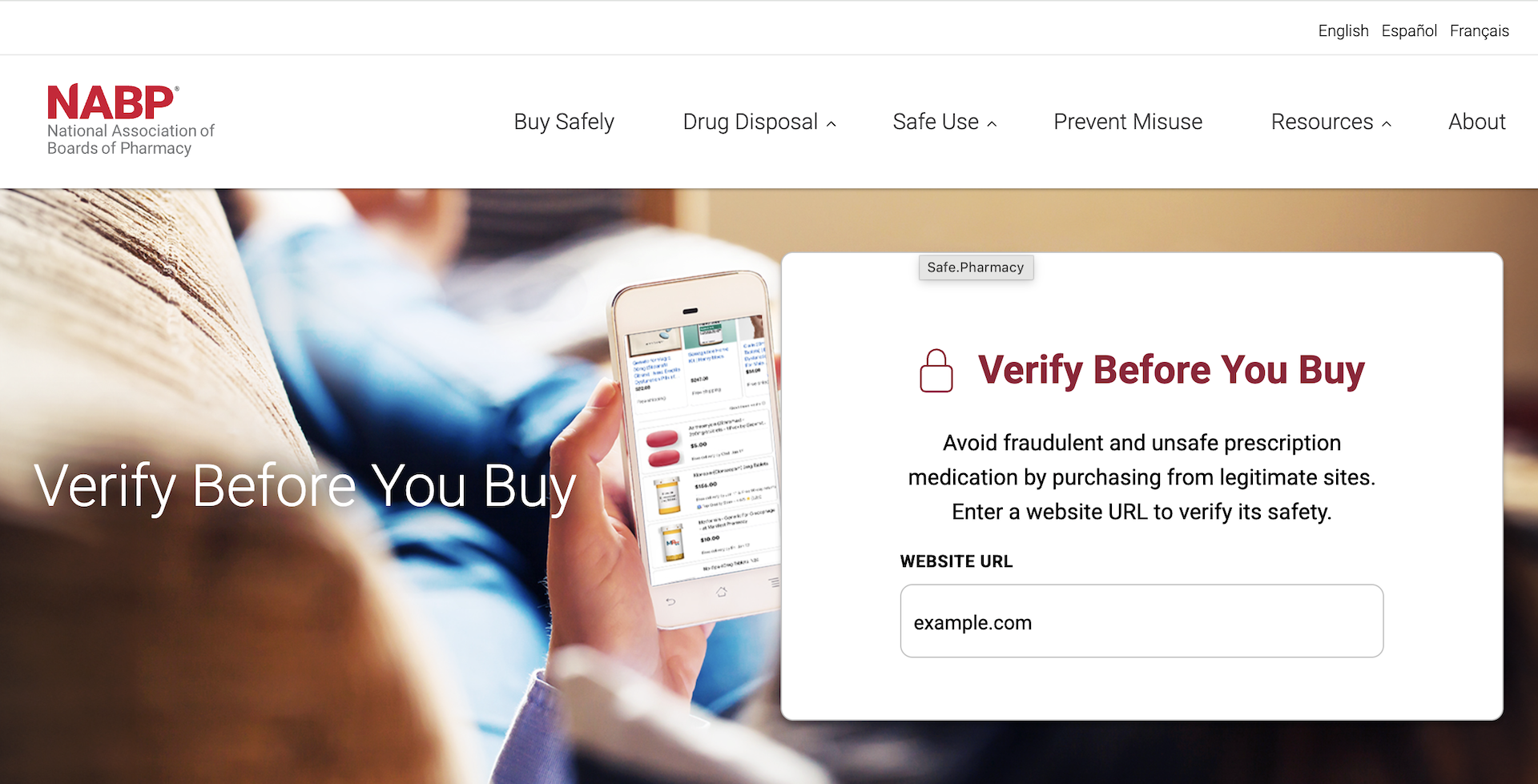
Verify Before You Buy Website
Avoid fraudulent and unsafe prescription medication by purchasing from legitimate sites. Search a website URL to verify its safety.

FDA Resources for Human Drug Compounding
FDA’s compounding program aims to protect patients from poor-quality compounded drugs, while preserving access to lawfully-marketed compounded drugs for patients who have a medical need for them.
The Alliance for Safe Biologic Medicines (ASBM) is a diverse coalition of stakeholders, including physicians, pharmacists, patient advocates, researchers, and biopharmaceutical manufacturers. Since 2010, ASBM has worked with regulators worldwide to shape policies that reflect the best interests of patients, ensuring access to lifesaving and life-enhancing medicines while fostering innovation in healthcare.